Psychotropic Drug Import License
Psychotropic Drug Import License
精神类进口准许证
Spiritual import permit background
The import and export permit for psychotropic substances refers to the State Food and Drug Administration to implement import and export supervision and management of psychotropic drugs that directly act on the central nervous system to stimulate or inhibit the use of continuous use, and issue permits for the import and export of psychotropic substances. License. Any unit that imports and exports psychotropic drugs (including psychotropic drug standards and reference materials) by any means of trade, the Customs and Excise Department of the State Food and Drug Administration needs to issue and affix the “Special Seal for the Approval of Import and Export of Special Drugs of the State Food and Drug Administration” The psychiatric drug import permit or the psychotropic drug export permit (hereinafter referred to as the psychotropic drug import and export permit), and the customs clearance and inspection procedures are carried out according to the regulations.
The general process of importing psychotropic drugs in our company:
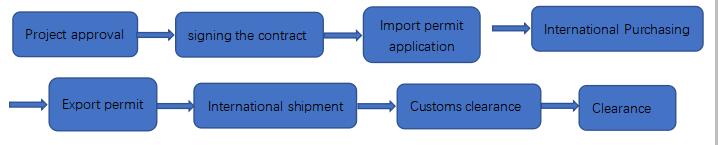
According to past experience, the requirements for the import of spiritual import permits are usually 4-6 weeks. Our company will provide customers with foreign notarization certificates, business licenses and product documents to assist customers in applying for import licenses. After successfully handling the spirit import license, our company will deliver the goods within 30 working days. Click to view the Spiritual Import License.
Applicants for psychotropic substances must pay attention to the following points:
1. The Psychotropic Drug Export Permit is valid for three months (the validity period does not span the year). The “Psychotropic Drug Import Permit” is valid for one year.
2. The import and export of psychotropic substances is permitted to confirm the “one batch one license” system, and the contents of the certificate cannot be changed by itself. If you need to change, you should go to the State Food and Drug Administration for a replacement procedure. The Customs examines and releases the import and export of psychotropic substances, and endorses and affixes the seals at the designated locations of the import and export permits for psychotropic substances.
3. Psychotropic drugs must be imported through ports approved by the State Council for importing drugs, and ports for importing psychotropic substances must be consistent with ports approved for the import of psychotropic drugs.
(1) Imported psychotropic ports: Beijing, Tianjin, Shanghai, Dalian, Qingdao, Chengdu, Wuhan, Chongqing, Xiamen, Nanjing, Hangzhou, Ningbo, Fuzhou, Guangzhou, The cities of Shenzhen, Zhuhai, Haikou, Xi’an and Nanning are directly under the jurisdiction of the Customs.
(2) For the first time, psychotropic drugs sold in China are limited to ports of three cities in Beijing, Shanghai and Guangzhou.
